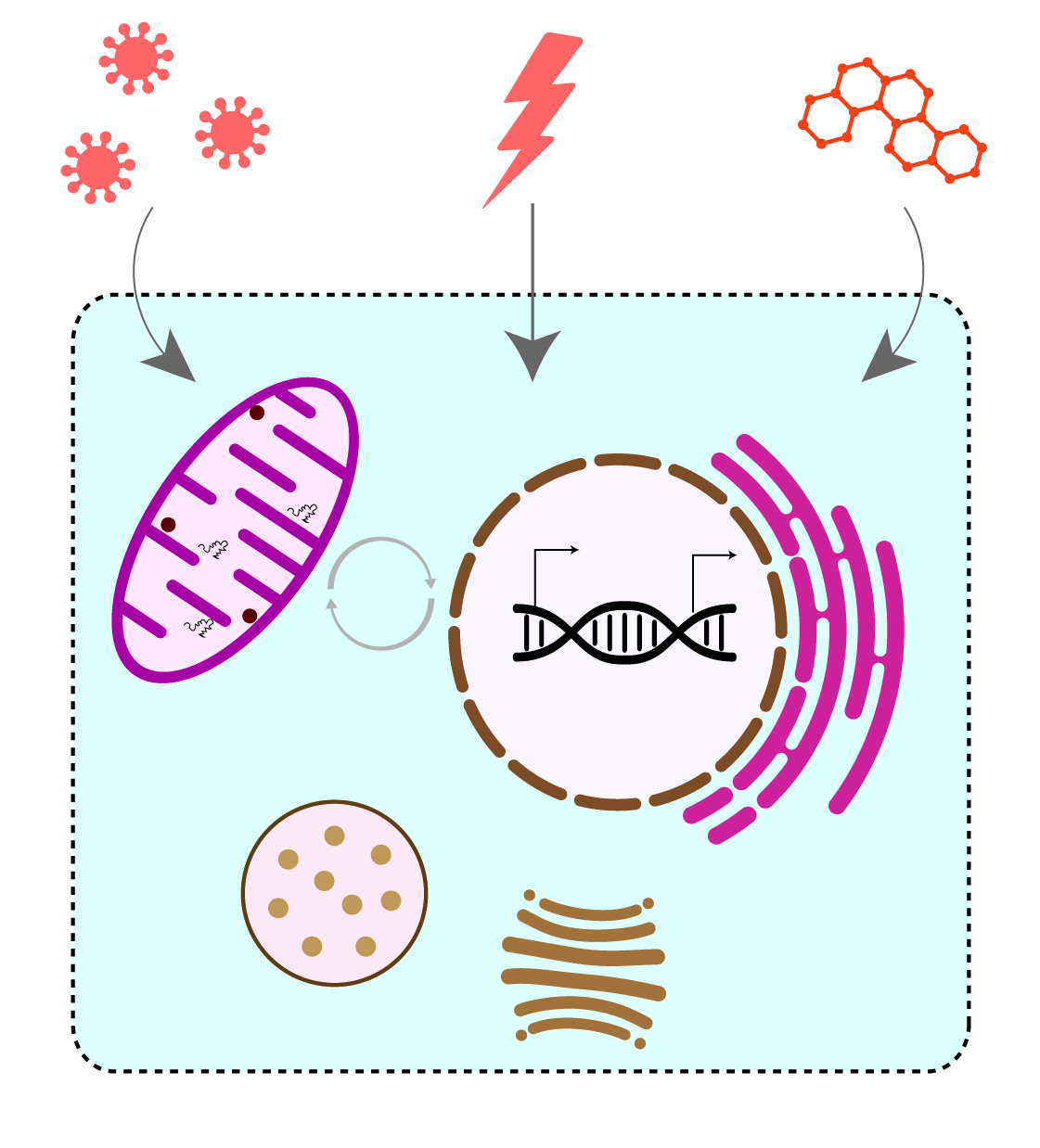
Cells continuously adapt to altering environmental conditions to maintain cellular homeostasis. External stimuli, such as small molecules, pathogenic bacteria and viruses, can trigger cellular stress responses. These dedicated pathways include specific molecular signaling pathways and organelle-specific protein quality control mechanisms to overcome damage.
RNF213 is a giant protein composed of a unique combination of AAA+ ATPase and E3 ubiquitin ligase. It is widely known for its affiliation with Moyamoya disease. Recently, RNF213 emerged as a novel player in host cell response to pathogenic stress. We are focusing on the characterization of the molecular and biochemical function of RNF213 to better understand its role in cellular homeostasis and disease.
Mitochondria, the central hub for cellular metabolism, plays a pivotal role in modulating stress signaling to maintain cellular homeostasis and dynamically adapt to changing energy requirements of the cell. Altered mitochondrial activity or mitochondrial dysfunction can trigger accumulation of senescent cells, which has severe implications in organismal ageing and age-associated diseases. However, the molecular signaling and regulatory events underlying mitochondrial stress induced senescence remain elusive. We aim to decipher the molecular mechanisms regulating mitochondrial stress signaling during senescence.
Mitochondrial RNA granules (MRG) are large membrane less structures essential for multiple steps of mitochondrial RNA metabolism. Despite the fact that mutations in components of those granules are linked to neurological disorders, the overall composition, structure and especially dynamic properties upon stress are unknown. We employ proximity labelling combined with quantitative mass spectrometry to elucidate the MRG architecture at high depth and to characterize their dynamic regulation upon stress.
The endoplasmic reticulum (ER) is the central hub for the secretion of proteins and faces a high amount of nascent proteins that require correct folding. We explore how cells adapt their ER protein quality control to various cellular stresses. Protein folding per se is error-prone and cells developed an elegant system for sensing and resolving accumulating amounts of unfolded and misfolded proteins, collectively known as the unfolded protein response (UPR). Mammalian cells contain three parallel UPR branches with different functions that cooperatively remodel the cellular proteome such that the folding deficit in the ER is overcome. We seek to understand how protein import at the ER is affected under stress conditions and define the contribution of the individual UPR branches in this scenario. By combining molecular, genetic and biochemical assays along with multi-layered systems biology, we aspire to unravel organellar and cellular stress signaling mechanisms to reveal their role in human diseases.