News from the Institute

Today, the German Research Foundation (DFG) announced that the transregional CRC on functionalisation of the ubiquitin system against cancer (UbiQancer) will be funded with € 18 M for the next four years. The CRC/TRR is coordinated by Technical University Munich (speaker: Prof. Florian Bassermann) with Goethe University Frankfurt and Julius-Maximilians-Universität Würzburg as co-applicant universities, and Universities of Kiel and Mainz, Helmholtz Munich, and the MPI of Biochemistry in Martinsried being involved as partners.
... (read more)
IBC2 Group leader Alexandra Stolz has been selected to become part of the AIM Council of International Rising Stars Program and an affiliate member of The AIM Center.
The Autophagy, Inflammation, and Metabolism Center of Biomedical Research (The AIM Center) is an NIH-funded center established on September 1st, 2017. The Center intends to promote autophagy research and international collaborative efforts, as well as help building the next generation of autophagy scientists.
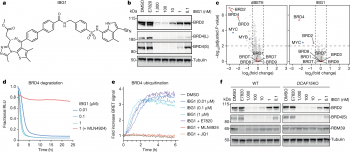
A new class of molecular glue could pave the way for a new generation of drugs to target cancers and neurodegenerative diseases.
... (read more)
A collaborative research project between scientists from Technion - the Israel Institute of Technology in Haifa, Goethe University Frankfurt and Helmholtz Center Munich/Ludwig Maximilian University Munich on “Understanding and Targeting Degradation-resistant Cancers” is funded with 1.6 million € as part of the German-Israeli Project Cooperation (DIP). DIP was inaugurated in 1997 by the German Federal Ministry for Education and Research (BMBF) to strengthen excellence in German-Israeli research cooperation. The Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) has taken over and continues the program while BMBF continues to provide funding for the five-year projects.
... (read more)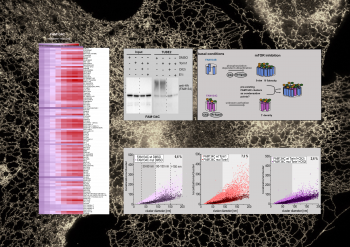
Content and dimensions of the endoplasmic reticulum (ER) are flexible and regulated based on cellular needs, environmental changes or stress. Turnover or degradation of superfluous material is facilitated by selective degradation of the ER via the lysosome (ER-phagy). Malfunction of ER-phagy usually results in disease formation, including neurodegenerative diseases.
... (read more)