22 Jul 2019 - Mechanism of Legionella toxicity control revealed
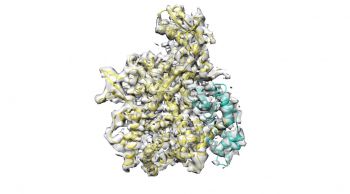
Legionella pneumophilia releases hundreds of enzymes that are instrumental for its propagation and subversion of the hosts’ immune system. Some of these virulence factors are extremely toxic, and their action needs to be strictly controlled to not kill host cells immediately. A team of scientists from IBC2 and EMBL Grenoble has now analyzed how Legionella keeps the family of SidE effectors in check and made a surprising discovery:
They found that a glutamylase activity in regulator SidJ does the job. SidJ attacks the central glutamate of SidE enzymes to attach chains of glutamate, thereby inhibiting SidE activity and limiting toxicity. So far, little is known about glutamylases. “Glutamylation as a protein modification is understudied. Our finding that bacteria uses glutamylation during infection certainly argues for more research in this field“, commented first author Sagar Bhogaraju, who is a former member of IBC2 and now leads a group at EMBL Grenoble. “This is a typical example of how completely unpredictable results drive research. Such discoveries are what make science a fascinating and exciting profession,” added IBC2 director Ivan Đikić, who led the interdisciplinary team. The researchers also revealed how SidJ is selectively activated in host cells: It requires the calcium-binding protein calmodulin which is present in mammalian cells. The now discovered mechanism opens up new possibilities for inhibiting the spread of Legionella in the host organism. “We’re currently working on selectively eliminating SidJ by developing inhibitors for the glutamylase domain. In addition to the use of antibiotics, they could prevent the spread of Legionella pneumophilia in phagocytes,” explains Đikić.