29 Mar 2016 - Shedding light on ALS: Novel mechanism in selective autophagy discovered.
ALS (Amyotrophic lateral sclerosis) is a rare and severe disease characterized by loss of motor neurons and neurodegeneration leading to death within 3-4 years.
The 2014 ice bucket challenge brought ALS to broader public attention but to date there is no treatment for ALS, despite intensive research in the field.
After being involved in the discovery of a kinase (Tank-binding kinase 1, TBK1) linked to ALS (Freischmidt el al., nature neuroscience 2015), researchers from the IBC2 in collaboration with international partners now succeeded in clarifying the physiological function of TBK1.
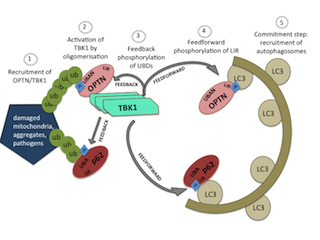
The group around Ivan Dikic revealed that TBK1 specifically phosphorylates its adaptor protein optineurin, thereby enabling a stronger binding of optineurin to ubiquitin that marks damaged mitochondria. By this means, TBK1 promotes an important cellular quality control system responsible for the clearance of damaged mitochondria and other cellular organelles, a process called selective autophagy.
Damaged mitochondria, TBK1 and optineurin have long been linked to neurodegenerative diseases such as Parkinson’s and ALS. However, the molecular details behind this connection are only beginning to be unveiled. Benjamin Richter, first author of the study, and his colleagues could further show that an ALS-associated TBK1 mutant fails to associate with optineurin and damaged mitochondria representing a possible link to the disease mechanism underlying ALS. The results are published in this weeks’ PNAS Online Early Edition.